A groundbreaking study published on December 19, 2025, in the esteemed journal Science Advances has unveiled a fundamental evolutionary trade-off that has profoundly shaped the trajectory of complex animal societies, particularly in ants. Researchers discovered that certain ant species have achieved massive colony sizes and remarkable evolutionary success by strategically de-prioritizing the individual robustness of their workers in favor of sheer numbers. This counter-intuitive approach—investing less in the protective body armor of each ant to produce more individuals—has provided critical insights into how the tension between quantity and quality can drive significant evolutionary outcomes, including the rise of intricate social structures observed in species from insects to humans.
The Enduring Biological Quandary: Quantity vs. Quality
The philosophical debate concerning the merits of quantity versus quality has long permeated various fields, from economics to ethics. In the realm of biology, this tension manifests as a fundamental evolutionary dilemma: should an organism invest heavily in producing a few highly resilient, well-equipped offspring, or should it prioritize the proliferation of numerous, albeit less robust, individuals? This question becomes even more complex when considering social organisms, where the individual’s role is intrinsically linked to the collective’s success. For decades, evolutionary biologists have grappled with understanding the mechanisms through which individual traits adapt and transform as societies become increasingly complex.
The recent research, spearheaded by lead author Arthur Matte, a Ph.D. student in zoology at the University of Cambridge, and senior author Evan Economo, chair of the Department of Entomology at the University of Maryland, directly addresses this quandary through an exhaustive analysis of ant species. Their findings suggest that for many successful ant lineages, the answer leaned decisively towards quantity, demonstrating that a "cheaper" individual, in terms of resource investment, can be an evolutionary advantage at the societal level.
Economo, who also holds the James B. Gahan and Margaret H. Gahan Professorship at UMD, articulated the core biological question driving this research: "There’s this question in biology of what happens to individuals as societies they are in get more complex. For example, the individuals may themselves become simpler because tasks that a solitary organism would need to complete can be handled by a collective." This concept of individual simplification within a complex social framework implies that organisms can become "cheaper" to produce, requiring fewer resources and enabling their production in larger quantities, even if each individual is less physically robust. Until now, this specific hypothesis had not been rigorously tested with large-scale analyses in social insects, making the current study a significant milestone.
Unpacking the Ant Paradox: From Individual Armor to Collective Might
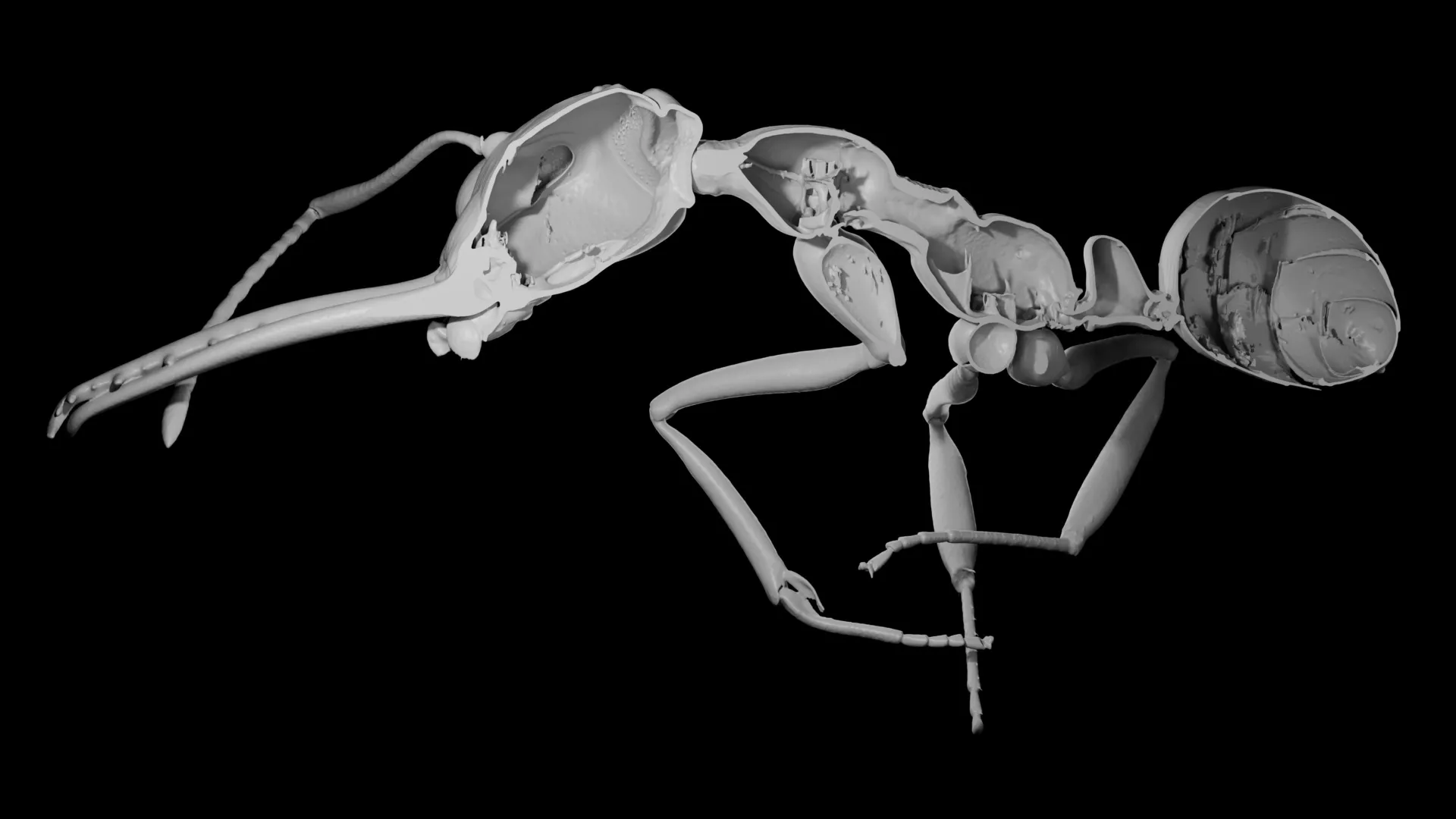
The study honed in on the ant’s cuticle—the hard, outer layer of its exoskeleton—as a key indicator of individual investment. The cuticle is a marvel of biological engineering, providing crucial protection against a myriad of environmental threats. It acts as a primary defense against predators, prevents desiccation in arid environments, and offers a barrier against pathogens and diseases. Beyond protection, it serves as the structural scaffold for muscles, enabling movement and supporting the ant’s overall body plan. However, this essential armor comes at a considerable biological cost.
Producing a robust cuticle demands significant investment in limited nutrients, particularly nitrogen and various minerals. These resources are finite, and their allocation towards a thicker, more durable cuticle directly reduces the resources available for other biological processes, including the production of more individuals. The research team hypothesized that a trade-off might exist: species that invest less in the individual worker’s cuticle could free up these valuable nutrients, redirecting them to generate a greater number of workers. This strategy, they posited, could be a driving force behind the formation of vast, complex ant colonies.
To test this intricate hypothesis, the researchers embarked on an ambitious data collection and analysis effort. They utilized an extensive dataset comprising 3D X-ray scans from more than 500 distinct ant species. This advanced imaging technique allowed them to meticulously measure both the total body volume and, crucially, the cuticle volume for each individual ant. The findings revealed a remarkable spectrum of cuticle investment, ranging from a mere 6% to a substantial 35% of an ant’s total body volume, highlighting the diverse evolutionary strategies employed across the ant kingdom. When these precise measurements were integrated into sophisticated evolutionary models, a distinct and compelling pattern materialized: ant species that demonstrated a lower relative investment in their cuticle consistently tended to form larger, more populous colonies. This finding provided robust empirical support for the proposed quantity-over-quality trade-off.
Methodology: Peering Inside Ant Architecture with Advanced Imaging
The success of this study relied heavily on its innovative and large-scale methodological approach. The use of 3D X-ray microtomography, a technique akin to a medical CT scan but at a microscopic resolution, allowed the researchers to non-invasively peer inside hundreds of ant specimens. This enabled them to create detailed three-dimensional models of each ant, from which precise volumetric measurements could be extracted. Such a comprehensive dataset, encompassing over 500 species, represents an unprecedented scale for studying morphological evolution in social insects.
The ability to accurately quantify cuticle volume relative to total body volume across such a diverse phylogenetic range was critical. It allowed the researchers to move beyond anecdotal observations and perform rigorous comparative analyses. By inputting these morphological data, alongside ecological and colony size information, into advanced phylogenetic comparative methods, they could reconstruct evolutionary trajectories and identify statistical correlations, thereby teasing out the causal links between individual investment, colony size, and diversification rates. This rigorous methodology underpins the strength and credibility of their conclusions, making the study a benchmark for future research in social insect evolution.
The Evolutionary Trade-Off: Cheaper Workers, Larger Societies
The core revelation of the study centers on the evolutionary success of producing "cheaper" workers. While individual ants with thinner cuticles are undoubtedly more vulnerable to direct threats—a fact Economo jovially referred to as the "evolution of squishability"—the research demonstrates that this individual fragility is more than compensated for at the collective level. Reduced investment in individual armor frees up valuable metabolic resources, allowing the colony to produce a greater number of workers. This numerical advantage then becomes the bedrock for a suite of highly effective social traits.
As Matte elaborated, "Ants reduce per-worker investment in one of the most nutritionally expensive tissues for the good of the collective. They’re shifting from self-investment toward a distributed workforce, resulting in more complex societies." This redistribution of resources from individual protection to collective strength fosters the development of highly organized and cooperative behaviors. These include more efficient cooperative foraging, where a larger workforce can exploit resources more effectively; enhanced shared nest defense, where overwhelming numbers can deter predators despite individual vulnerability; and a more pronounced division of labor, allowing for greater specialization and overall colony efficiency. These collective traits become increasingly sophisticated and effective as colony sizes grow, creating a positive feedback loop that reinforces the initial trade-off.
A Catalyst for Diversification: How "Squishability" Fuels Speciation
Perhaps one of the most striking findings of the study is the unexpected link between lower investment in cuticle and higher diversification rates. Biologists often consider diversification—the frequency at which new species arise within a lineage—as a key metric of evolutionary success. Economo noted the rarity of identifying specific traits directly correlated with diversification in ants, making this particular result exceptionally noteworthy.
The exact mechanisms through which reduced cuticle investment promotes speciation are still being explored, but the researchers propose compelling hypotheses. One leading idea posits that ants with lower nutritional demands, due to their less resource-intensive body plans, gain a significant ecological advantage. They become more versatile and adaptable, capable of colonizing and thriving in environments where resources, particularly nitrogen, are limited. As Matte explained, having "less nitrogen could make them more versatile and able to conquer new environments." This enhanced ecological flexibility could drive geographical expansion and subsequent reproductive isolation, leading to the formation of new species. Matte initiated this foundational work during his master’s program while interning in Economo’s lab at the Okinawa Institute of Science and Technology in Japan, highlighting the long-term nature of this scientific endeavor.
Furthermore, the authors suggest a reinforcing cycle: as ant societies evolved greater complexity, group-level defenses—such as highly organized nest protection, sophisticated alarm systems, and collective disease control mechanisms—began to mitigate the necessity for heavy individual armor. This reduced pressure on individual ants to be heavily protected then allowed for further reductions in cuticle investment, which, in turn, facilitated even larger colonies. This self-perpetuating cycle could have accelerated both the growth of complex societies and the rates of speciation.
Ants as a Model System: A Window into Social Evolution
Ants are not merely fascinating creatures; they represent an extraordinarily powerful model system for studying the intricacies of social evolution. Their global distribution, immense species diversity (over 14,000 described species), and wide range of colony sizes—from a mere few dozen individuals to multi-million member supercolonies—provide a natural laboratory for evolutionary biologists. This vast spectrum of social organization allows researchers to observe and analyze different evolutionary strategies and their outcomes under varying ecological pressures.
As lead author Arthur Matte highlighted, "Ants are everywhere. Yet the fundamental biological strategies which enabled their massive colonies and extraordinary diversification remain unclear." This study addresses a critical gap in our understanding, demonstrating how a seemingly minor change at the individual level—cuticle thickness—can ripple through an entire social system, influencing its size, complexity, and ultimately, its evolutionary success. The insights gained from ants are often highly transferable to other social organisms, including termites, certain bees, and even, as the study suggests, humans.
Echoes Across Evolution: From Multicellularity to Human Societies
The implications of this research extend far beyond the insect world, offering profound parallels to fundamental evolutionary transitions and the development of complex systems throughout biological history. The researchers draw a direct analogy to the evolution of multicellularity, a pivotal moment in life’s history where individual, solitary cells began to cooperate, forming larger, more complex organisms. In this context, individual cells within a multicellular organism are often simpler and less independently robust than their free-living unicellular ancestors, yet collectively, they achieve a far greater level of complexity and functional specialization. Matte eloquently stated, "It’s a pattern that echoes the evolution of multicellularity, where cooperative units can be individually simpler than a solitary cell, yet collectively capable of far greater complexity."
The findings also resonate deeply with aspects of human societal development and military history. Economo pointed to the historical shift in warfare from heavily armored individual combatants, like medieval knights, to more specialized and numerous forces, such as archers and crossbowmen, who, while individually less protected, could collectively overwhelm an opponent. He also invoked Lanchester’s Laws, mathematical equations developed during World War I that precisely quantify the advantages of numerical superiority in combat, even when individual fighters are less powerful. These historical and mathematical parallels underscore the universality of the quantity-versus-quality trade-off in complex systems.
Matte summarized the broader significance, stating, "The tradeoff between quantity and quality is all around. It’s in the food you eat, the books you read, the offspring you want to raise. It was fascinating to retrace how ants handled it through their long evolution. We could see lineages taking different directions, being shaped by different constraints and environments, and ultimately giving rise to the extraordinary diversity we observe today." This perspective highlights the study’s contribution to understanding fundamental principles that govern the organization and evolution of life itself.
Future Directions and Unanswered Questions
While this study provides groundbreaking insights, it also opens numerous avenues for future research. One immediate question is whether similar evolutionary paths have been followed by other social organisms. The authors specifically mention termites as a potential candidate, noting that this possibility requires further rigorous testing. Investigating the genetic and developmental mechanisms underlying cuticle thickness variation would also provide a deeper understanding of the molecular basis of this evolutionary trade-off.
Furthermore, exploring the precise ecological conditions and selective pressures that favor the "quantity" strategy over "quality" in different ant lineages could yield valuable insights into niche specialization and adaptive radiation. Understanding how various environmental factors, such as predator prevalence, resource availability, and climate, interact with this trade-off will be crucial for a comprehensive picture of ant evolution. The study’s findings also prompt further investigation into the intricate interplay between individual morphology, collective behavior, and the rates of speciation, potentially revealing general principles applicable across the tree of life.
The paper, titled "The evolution of cheaper workers facilitated larger societies and accelerated diversification in ants," represents a monumental step forward in our understanding of social evolution. It underscores how seemingly small, individual-level adaptations can cascade into profound societal transformations and drive the astonishing diversity of life on Earth.
This research was made possible through the generous support of the Okinawa Institute of Science and Technology, the Japan Society for the Promotion of Science KAKENHI (24K01785), the University of Cambridge, and the General Research Fund 2022/2023 (17121922) from the Research Grant Council of Hong Kong. It is important to note that the views expressed in this article do not necessarily reflect the official positions of these funding organizations.