The age-old philosophical tension between quantity and quality, often debated in human endeavors, has now been revealed as a fundamental driver shaping the very fabric of evolution, particularly in the emergence of complex animal societies. New research, published on December 19, 2025, in the esteemed journal Science Advances, offers compelling evidence that ant species have harnessed this trade-off, opting for numerical superiority over individual robustness to achieve remarkable evolutionary success. This pivotal study suggests that by investing less in the protective outer layer of individual workers, ant colonies unlock resources to produce more individuals, fostering larger societies and accelerating the formation of new species.
The Strategic Retreat from Individual Toughness
At the heart of this groundbreaking discovery lies the ant cuticle – the hard, chitinous exoskeleton that serves as both armor and structural support. Historically, a thicker, more robust cuticle has been perceived as an evolutionary advantage, protecting individual insects from predators, desiccation, and disease. However, the research, led by senior author Evan Economo, chair of the Department of Entomology at the University of Maryland, and lead author Arthur Matte, a Ph.D. student in zoology at the University of Cambridge, unveils a counter-intuitive strategy. They found that certain ant species have evolved to prioritize the production of a greater number of less-protected workers over a smaller contingent of heavily armored ones. This strategic reallocation of resources, particularly valuable nutrients like nitrogen and various minerals, allows colonies to produce a significantly larger workforce.
"There’s this question in biology of what happens to individuals as societies they are in get more complex. For example, the individuals may themselves become simpler because tasks that a solitary organism would need to complete can be handled by a collective," Economo explained, highlighting the core hypothesis that individuals within complex societies might become "cheaper" – requiring fewer resources to build and thus producible in greater numbers, even if physically less robust. This concept of "cheaper" individuals, a cornerstone of the study, had lacked explicit, large-scale empirical validation in social insects until now.
Ants as a Paradigm for Social Evolution
Ants, with their extraordinary diversity and ubiquitous presence across nearly every terrestrial ecosystem, offer an unparalleled natural laboratory for studying the intricacies of social evolution. With over 15,000 known species and subspecies, ranging from small, solitary foragers to vast mega-colonies housing millions, ants exhibit an astonishing spectrum of social organization. This variability in colony size and structure, coupled with their deep evolutionary history spanning over 100 million years, makes them an ideal model system to investigate the drivers of social complexity. Their eusocial nature – characterized by cooperative brood care, overlapping generations within a colony, and a division of labor into reproductive and non-reproductive castes – represents one of the pinnacles of evolutionary cooperation.
Despite their ecological dominance and profound impact on ecosystems globally, the fundamental biological strategies underpinning their massive colonies and extraordinary diversification have remained largely enigmatic. "Ants are everywhere," Matte noted, underscoring the paradox that while their success is undeniable, the specific evolutionary mechanisms enabling it have been less clear. The research team specifically hypothesized that a crucial link might exist between colony size and the metabolic investment an ant species makes in its individual workers’ cuticles.
The Biomechanical and Nutritional Cost of Body Armor
The insect cuticle is far more than just a shell; it is a complex, multi-layered biomaterial vital for survival. Composed primarily of chitin, a tough polysaccharide, embedded in a matrix of proteins and lipids, the cuticle provides crucial functions:
- Protection: It acts as a primary defense against mechanical injury, desiccation (water loss), and pathogen invasion.
- Structural Support: It provides the rigid framework for muscle attachment, enabling movement and locomotion.
- Sensory Interface: It houses various sensory organs, allowing ants to interact with their environment.
However, constructing and maintaining this intricate biological armor comes at a significant metabolic cost. The synthesis of chitin and its associated proteins requires a substantial allocation of limited nutrients, particularly nitrogen, which is often a bottleneck resource in many ecosystems. Minerals like calcium and magnesium are also incorporated to enhance rigidity. Producing a thicker, more resilient cuticle demands a greater share of these precious resources, which in turn limits the total number of individuals a colony can sustain. This inherent trade-off forms the crux of the research’s hypothesis: colonies face a fundamental decision on how to allocate resources – either towards fewer, more robust individuals or a greater multitude of less individually fortified ones.
Unveiling Evolutionary Trends Through Advanced Imaging
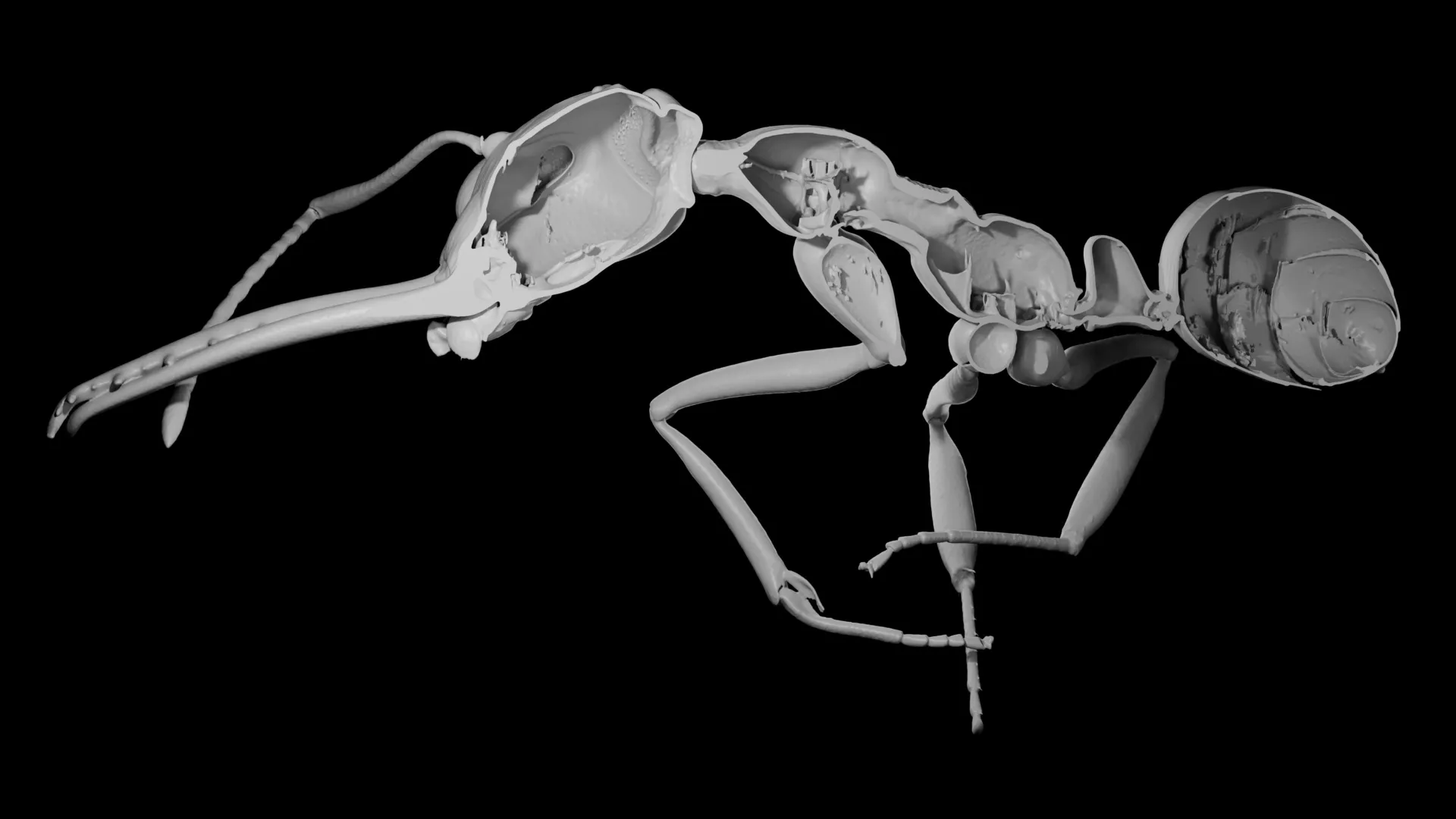
To rigorously test their hypothesis, the research team embarked on an ambitious comparative study, analyzing an extensive dataset comprising 3D X-ray scans from over 500 distinct ant species. This involved utilizing advanced micro-computed tomography (micro-CT) techniques, which allowed them to non-invasively visualize and quantify the internal and external morphology of individual ants with remarkable precision. For each specimen, researchers meticulously measured both the total body volume and, critically, the specific volume occupied by the cuticle.
The analysis revealed a wide spectrum of cuticle investment across species, ranging from a mere 6% to a substantial 35% of an ant’s total body volume. This significant variation immediately suggested an underlying evolutionary flexibility in how different species allocated resources to their exoskeletons. When these precise morphological measurements were integrated into sophisticated evolutionary models, a clear and consistent pattern emerged: ant species that demonstrated a lower relative investment in their cuticle consistently tended to form larger colonies. This robust correlation provided strong empirical support for the "cheaper worker" hypothesis.
The Collective Strength Paradigm: More Than the Sum of Its Parts
While the findings indicate that thinner cuticles render individual ants more vulnerable to environmental stressors and predators, the authors propose that this apparent individual disadvantage is cleverly offset, and indeed leveraged, by the advantages inherent in large, complex societies. The reduction in individual armor investment appears to be intrinsically linked to the development and enhancement of other crucial social traits, including:
- Cooperative Foraging: A larger workforce can more efficiently discover, exploit, and transport food resources back to the nest.
- Shared Nest Defense: Even individually "squishier" ants can mount a formidable collective defense when acting in concert, overwhelming predators through sheer numbers and coordinated attacks.
- Division of Labor: Larger colonies can develop highly specialized castes and roles, leading to increased efficiency in tasks such as brood care, nest maintenance, and resource processing.
These collective traits, which tend to become more pronounced and effective as colony size increases, effectively compensate for the reduced physical robustness of individual workers. As Arthur Matte elucidated, "Ants reduce per-worker investment in one of the most nutritionally expensive tissues for the good of the collective. They’re shifting from self-investment toward a distributed workforce, resulting in more complex societies." This mirrors a profound evolutionary principle observed in the transition from solitary life forms to multicellular organisms, where individual cells, though simpler than their free-living ancestors, achieve far greater complexity and functionality when integrated into a cooperative whole.
A Catalyst for Diversification: Why Less Armor Means More Species
Perhaps one of the most striking findings of the study was the observed link between lower cuticle investment and higher diversification rates. In evolutionary biology, diversification – the frequency at which new species arise – is a critical indicator of evolutionary success. Economo highlighted the significance of this result, noting that very few specific traits have been definitively connected to diversification rates in ants, making this particular finding especially noteworthy.
The precise mechanisms by which reduced cuticle investment promotes speciation are still subjects of ongoing investigation, but the researchers propose several compelling hypotheses. One leading idea posits that ants with lower nutritional demands, particularly for nitrogen-rich cuticle components, gain a significant ecological advantage. They become more metabolically efficient and less constrained by resource availability, enabling them to expand into novel environments where resources might be limited or unpredictable. "Requiring less nitrogen could make them more versatile and able to conquer new environments," Matte suggested, linking metabolic flexibility to ecological adaptability and, ultimately, to the opening of new evolutionary niches.
Furthermore, the authors suggest a reinforcing feedback loop: as ant societies become increasingly complex and reliant on group-level defenses (e.g., collective nest protection, sophisticated disease control mechanisms within the colony), the selective pressure for heavily armored individual workers diminishes. This reduction in individual armor, in turn, facilitates the production of larger colonies, which further strengthens collective defenses and reduces the need for individual robustness. This continuous cycle likely drove the evolutionary trajectory towards "squishier" but more numerous workers. Economo humorously captured this phenomenon, stating, "I think of this as the evolution of squishability. Many kids have discovered that insects aren’t all equally robust." While the study focused on ants, the researchers acknowledge the possibility that other social organisms, such as termites, may have followed similar evolutionary pathways, a prospect that warrants further dedicated research.
Echoes in Human History: Lessons from the Ant Colony
The implications of these findings extend far beyond the realm of entomology, offering profound insights into the fundamental principles governing the evolution of complexity, including those observed in human societies. The researchers draw compelling parallels to human military history, where the shift from individually formidable, heavily armored knights to specialized, lighter, and more numerous soldiers (like archers and crossbowmen) marked a significant strategic evolution. This historical transformation reflects the same quantity-quality trade-off observed in ant evolution.
Economo further pointed to Lanchester’s Laws, a set of mathematical equations developed during World War I to model combat dynamics. These laws demonstrate how, under certain conditions, a larger force of individually weaker fighters can overpower a smaller force of stronger, more robust adversaries. The ant study provides a striking biological validation of these principles, illustrating how numerical superiority, even with individually less potent units, can be a dominant evolutionary strategy.
"The tradeoff between quantity and quality is all around. It’s in the food you eat, the books you read, the offspring you want to raise," Matte eloquently summarized, underscoring the universality of this evolutionary dilemma. "It was fascinating to retrace how ants handled it through their long evolution. We could see lineages taking different directions, being shaped by different constraints and environments, and ultimately giving rise to the extraordinary diversity we observe today." This research not only illuminates a critical chapter in ant evolution but also offers a powerful lens through which to understand the broader forces that drive the emergence of complex life and social structures across the tree of life.
The paper, "The evolution of cheaper workers facilitated larger societies and accelerated diversification in ants," published in the journal Science Advances on December 19, 2025, represents a significant contribution to our understanding of social evolution and resource allocation. This research was generously supported by the Okinawa Institute of Science and Technology, the Japan Society for the Promotion of Science KAKENHI (24K01785), the University of Cambridge, and the General Research Fund 2022/2023 (17121922) from the Research Grant Council of Hong Kong. It is important to note that this article does not necessarily reflect the views of these funding organizations.